August 13, 2019 | Jola Glotzer
3D structure of hepatitis A virus IRES: a three-way junction
A CBC Lever Award Pilot Project awarded to Joseph Piccirilli, UChicago, resolved the 3D structure of internal ribosomal entry sites (IRES) of picornaviral RNA
Congratulations to Joe Piccirilli, UChicago, senior and corresponding author on an August 9th publication in Nature Communications, titled “A conserved RNA structural motif for organizing topology within picornaviral internal ribosome entry sites.” As reported in the Acknowledgements, “This work was supported by grants from the National Institutes of Health (…) and the Chicago Biomedical Consortium with support from the Searle Funds at The Chicago Community Trust to Joseph A. Piccirilli.” The CBC support came from a Pilot Project awarded to Piccirilli that was funded by a 2012 CBC Lever Award, “Synthetic Antibody Consortium,” and titled “Production of High Affinity sABS Against RNA and RNP Targets: Library Development and Chaperone Assisted Crystallography.”
The current Nature Communications publication reports on using chaperone assisted crystallography to resolve the 3D structure of hepatitis A virus (HAV) IRES domain at 2.84-Å resolution. In this research, the chaperone is a synthetic antibody fragment that binds to and stabilizes the viral RNA IRES domain.
The authors report that the HAV IRES domain organizes itself into a three-way junction, presumably a structural configuration that precedes its engagement with the host’s translational machinery. The HAV IRES shares topological similarities with two other members of the picornaviridae family — viruses causing encephalomyocarditis and foot-and-mouth disease, respectively — supporting a hypothesis that this structurally-conserved RNA motif is important for the initiation of translation.
Phoebe Rice, UChicago, is second senior author on the publication. Rice also has ties to the CBC; in 2016, together with a postdoc in her lab, Ignacio Mir Sanchis, she received a CBC Postdoctoral Research Award and in 2013, a CBC Catalyst Award.
Congratulations to all authors contributing to the current study!
Publication attributed to the CBC funding*:
Koirala D, Shao Y, Koldobskaya Y, Fuller JR, Watkins AM, Shelke SA, Pilipenko EV, Das R, Rice PA, Piccirilli JA. A conserved RNA structural motif for organizing topology within picornaviral internal ribosome entry sites. Nat Commun. 2019 Aug 9;10(1):3629. (PubMed)
ABSTRACT:
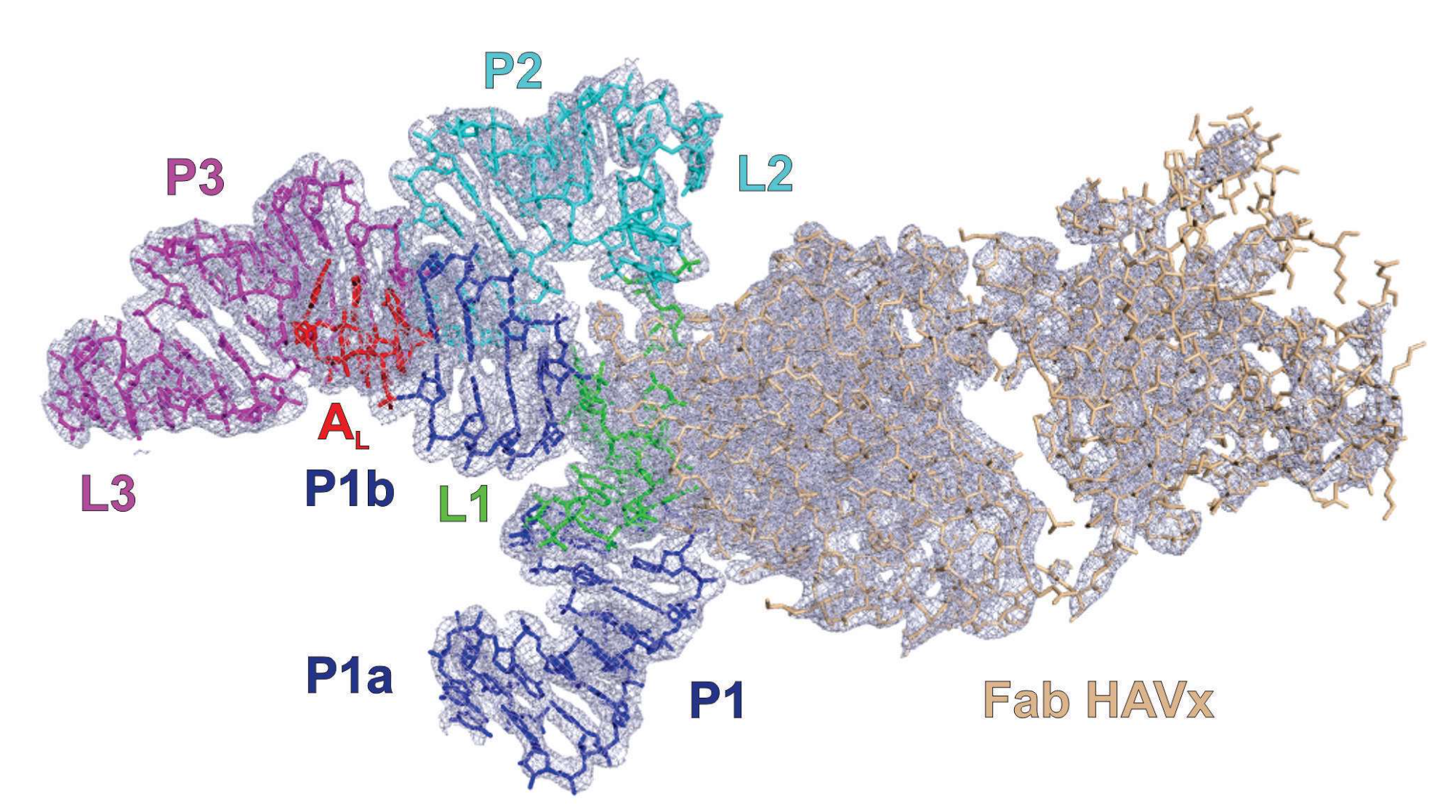
Crystal structure of HAV IRES dV RNA in complex with Fab HAVx solved at 2.84-Å resolution. Blue mesh represents the 2|Fo| – |Fc| electron density map at 1σ contour level and carve radius 1.8 Å. (Source: www.nature.com)
Picornaviral IRES elements are essential for initiating the cap-independent viral translation. However, three-dimensional structures of these elements remain elusive. Here, we report a 2.84-Å resolution crystal structure of hepatitis A virus IRES domain V (dV) in complex with a synthetic antibody fragment—a crystallization chaperone. The RNA adopts a three-way junction structure, topologically organized by an adenine-rich stem-loop motif. Despite no obvious sequence homology, the dV architecture shows a striking similarity to a circularly permuted form of encephalomyocarditis virus J-K domain, suggesting a conserved strategy for organizing the domain architecture. Recurrence of the motif led us to use homology modeling tools to compute a 3-dimensional structure of the corresponding domain of foot-and-mouth disease virus, revealing an analogous domain organizing motif. The topological conservation observed among these IRESs and other viral domains implicates a structured three-way junction as an architectural scaffold to pre-organize helical domains for recruiting the translation initiation machinery.
ACKNOWLEDGMENTS:
This work was supported by grants from the National Institutes of Health (R01AI081987, R01GM102489) and the Chicago Biomedical Consortium with support from the Searle Funds at The Chicago Community Trust to Joseph A. Piccirilli. The crystallographic work is based on research conducted at the Advanced Photon Source on the Northeastern Collaborative Access Team beamlines, 24-ID-B and 24-ID-C, which is supported by a grant from the National Institute of General Medical Sciences (P41 GM103403) from the National Institutes of Health. This research used resources of the Advanced Photon Source, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357. For SAXS data collection, this work used Advanced Light Source beamline SIBYLS, a US DOE Office of Science User Facility operated for the DOE Office of Science by Lawrence Berkeley National Laboratory under Integrated Diffraction Analysis (IDAT) grant contract DE-AC02-05CH11231. We would like to thank staffs of the Advanced Photon Source at Argonne National Laboratory and Advanced Light Source beamline SIBYLS for providing technical advice during data collection. We are thankful to Dr. Engin Ozkan, University of Chicago for helping with X-ray data processing. We also would like to thank Piccirilli laboratory members, especially to Benjamin Weissman, for critical review of the manuscript.
Featured CBC Community member(s):
Joseph Piccirilli, UChicago
- CBC Catalyst Award (2015):
▸ Toward the Visualization of Spliceosomal Intermediates
PIs: Yuan He (NU), Joseph Piccirilli (UChicago) and Jonathan Staley (UChicago) - *CBC Lever Award Pilot Project (2014, renewed for 2015):
▸ Lever-CSAP Pilot Project: Production of High Affinity sABS Against RNA and RNP Targets: Library Development and Chaperone Assisted Crystallography
PIs: Joseph Piccirilli (UChicago) and Alfonso Mondragon (NU)
Phoebe Rice, UChicago
- CBC Postdoctoral Research Award (2016):
▸ Cryo-EM Analysis of the Initiation of Replication of Two Mobile Elements Linked to Disease
PIs: Ignacio Mir Sanchis (postdoc) and Phoebe Rice (mentor; UChicago) - CBC Catalyst Award (2013):
▸ Structural Basis for RECA Filament Nucleation by the Neisseria Pile G Quadruplex Motif
PIs: Phoebe Rice (UChicago) and H. Steven Seifert (NU)
Articles published in the past about the featured CBC community members:
November 25, 2013
▸ Life on Earth Began in a World Based Solely on RNA

