February 5, 2019 | Jola Glotzer
Macrophages – a dance of life with death
Five (!) CBC community members collaborate on how to speed up heart recovery after an infarct
This could be truly called an exemplary CBC collaboration! An NU collaboration including four CBC awardees (Edward Thorp, Navdeep Chandel, David Gius, and Paul Schumacker), CBC event participants (Edward Thorp and Matthew Schipma) and even one CBC board member (Navdeep Chandel), co-authored a recent publication in Cell Metabolism, titled “Efferocytosis Fuels Requirements of Fatty Acid Oxidation and the Electron Transport Chain to Polarize Macrophages for Tissue Repair.” Thorp, who is senior author on the paper, has acknowledged partial funding of the published work by his CBC Catalyst Award (2013). The paper describes a novel mechanism that appears to involve macrophages which, upon digestion of damaged heart cells, cool off the local inflammation processes and activate a repair mechanism. The metabolites of the digested cells seem to play an important role in this transition. More is to be done to elucidate the exact mechanism, but the process might be general and applicable to any tissue injury. CBC congratulates all authors on publishing these exciting results!
Scientists Discover New Heart Attack Repair Pathway
Northwestern Medicine News | by Anna Williams | January 2, 2019
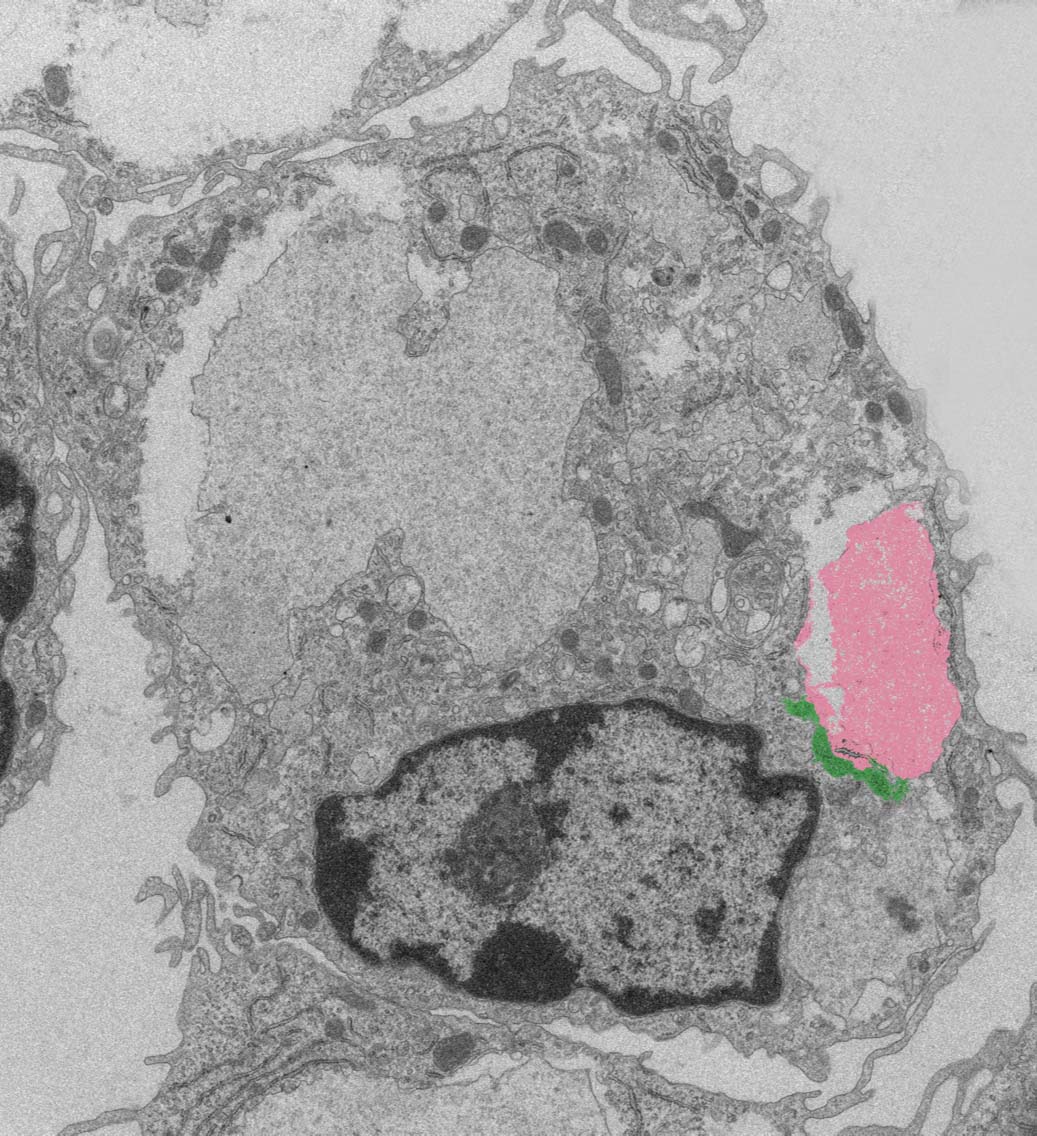
A macrophage immune cell, with a dead cell (pink) that has been eaten, and a mitochondrion (green) between the dead cell and the nucleus. The study’s findings indicate that what the macrophage eats is taken up by the mitochondrion, which in turn communicates with the nucleus to activate the macrophage to promote tissue repair.
Northwestern Medicine scientists have discovered a novel signaling pathway that promotes healing after a heart attack. The study, published in Cell Metabolism, demonstrates for the first time that the metabolism of dead heart cells by macrophages reprograms the immune cells to launch an anti-inflammatory response and stimulate tissue repair.
“Targeting this pathway may have therapeutic benefit for a broad range of acute and chronic disorders — not only heart attacks — by enhancing the active resolution of inflammation,” said principal investigator Edward Thorp, PhD, associate professor of Pathology and a member of the Feinberg Cardiovascular and Renal Research Institute (FCVRI).
Shuang Zhang, PhD, who recently graduated from Feinberg’s Driskill Graduate Program in Life Sciences (DGP), was the first author.
The Thorp Laboratory studies how immune cells regulate wound healing, in particular after myocardial infarction, or heart attack. After a patient has experienced a heart attack, immune cells are essential for removing dead or dying heart cells. If the cells aren’t cleared efficiently, they can lead to prolonged inflammation and cause further damage to the heart.
There are currently few effective treatments to enhance the healing process after a heart attack, and many patients go on to develop heart failure as a result of the damage.
The Thorp Laboratory has published extensively on efferocytosis — the process by which immune cells called macrophages engulf and ingest dying cells — and its critical role in turning on inflammation resolution after heart attack. But the intracellular mechanisms of the process remained unclear.

Principal investigator Edward Thorp, PhD, associate professor of Pathology and a member of the Feinberg Cardiovascular and Renal Research Institute, and first author Shuang Zhang, PhD, a recent graduate of Feinberg’s Driskill Graduate Program in Life Sciences.
In the new research, the scientists discovered that when macrophages swallow dead heart cells during efferocytosis, the macrophages fill with metabolites that reprogram the cell to initiate an anti-inflammatory response and promote repair of the heart.
“Our findings elucidate a novel process by which dead tissue metabolites are recycled to fuel wound healing,” Thorp explained. “It extends the meaning of ‘you are what you eat’ to the cellular level.”
In an experimental model, the scientists further demonstrated that when mitochondria — an organelle which regulates metabolism — are defective, healing after cardiac injury is impaired.
The findings suggest a novel role for immune cell metabolism beyond the production of energy.
“The idea that mitochondria, or overall metabolism, can act as a dictator of the cell and control macrophage function — that’s very novel,” Zhang said.
The Thorp Laboratory is continuing to investigate metabolic control of human immune cell function in further studies of cardiac injury and organ transplantation. Zhang is beginning a post-doctoral fellowship at Harvard Medical School, where she will continue her study of immune cells.
Other co-authors of the study include Navdeep Chandel, PhD, the David W. Cugell, MD, Professor of Medicine in the Division of Pulmonary and Critical Care and Cell and Molecular Biology; David Gius, MD, PhD, professor of Radiation Oncology and Pharmacology; Paul Schumacker, PhD, professor of Pediatrics in the Division of Neonatology, of Medicine in the Division of Pulmonary and Critical Care, and of Cell and Molecular Biology; Issam Ben-Sahra, PhD, assistant professor of Biochemistry and Molecular Genetics; Matthew DeBerge, PhD, research assistant professor of Pathology; Matthew Schipma, PhD, research assistant professor of Biochemistry and Molecular Genetics; Samuel Weinberg, a MD/PhD student in the Medical Scientist Training Program; and collaborators at Washington University in St. Louis and INSERM in France.
Thorp, Chandel, Gius, Schumacker and Ben-Sahra are also members of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University.
The study was supported by the National Heart, Lung, and Blood Institute F32HL127958, HL122309 and HL139812, Chicago Biomedical Consortium, American Heart Association predoctoral fellowship, Northwestern University presidential fellowship, and Sidney Bess Memorial Fund.
Source:
Adapted (with modifications) from the Northwestern Medicine News by Anna Williams, published on January 2, 2019.
Publication attributed to CBC funding*:
Zhang S, Weinberg S, DeBerge M, Gainullina A, Schipma M, Kinchen JM, Ben-Sahra I, Gius DR, Charvet LY, Chandel NS, Schumacker PT, Thorp EB. Efferocytosis Fuels Requirements of Fatty Acid Oxidation and the Electron Transport Chain to Polarize Macrophages for Tissue Repair. Cell Metab. 2018 Dec 20. [Epub ahead of print] (PubMed)
Featured scientist(s) with ties to cbc:
Edward Thorp, NU
- *CBC Catalyst Award (2013):
▸ APOA-I Mediated Regulation of Inflammation and Heart Repair after Heart Attack
PIs: Godfrey S. Getz (UChicago) and Edward Thorp (NU) - CBC Exploratory Workshop (2013):
▸ The CBC Exploratory Workshop on Lipoproteins
Edward Thorp (NU) – Workshop Speaker
Navdeep Chandel, NU
- CBC HTS Award (2015):
▸ Discovering Small Molecules to Improve Mitochondrial Function for Neuron Protection
PIs: Inma Martinez-Reyes and Navdeep Chandel (NU) - CBC Catalyst Award (2011):
▸ Oxygen Sensing in Endothelial Progenitor Cells
PIs: Jalees Rehman (UIC), David Eddington (UIC) and Navdeep Chandel (NU) - CBC Catalyst Review Board (CRB) (2014-2018):
▸ CRB Current Members
Navdeep Chandel (NU) — Board Member
David Gius, NU
- CBC Catalyst Award (2015):
▸ Fluorescent Probes for Live-cell Imaging of Sirtuin Activity: Application to Breast Cancer
PIs: Terry Vanden Hoek (UIC) and Alan Leff (UChicago)
Paul Schumacker, NU
- CBC Catalyst Award (2009):
▸ New Molecular Tools to Study Cancer Metabolic Reprogramming
PIs: Sergey Kozmin (UChicago) and Paul Schumacker (NU) - CBC Catalyst Award (2007):
▸ Proteomic Analysis of Mitochondrial and Sarcomeric Proteins in Cardiomyopathy
PIs: Elizabeth McNally (NU; at UChicago then), Paul Schumacker (NU), R. John Solaro (UIC), Hossein Ardehali (NU)
Matthew Schipma, NU
- CBC Tech Day (2014):
▸ Cutting-Edge Technologies — Driving Science Forward
Matthew Schipma — Panelist
related:
October 12, 2017
success story
▸ Mending a damaged heart is close-to-heart for CBC researcher Edward Thorp
